Aortic Aneurysm and Dissection
Important information regarding resolution and images. If you are experiencing issues with shifting of the pictures on the pages, you will need to change your browser from 100% to 90%.


Objectives - Topics to be Covered
-
Anatomy
-
When to Operate: Aortic Aneurysm vs Aortic Dissection
-
Procedure
-
Cannulation strategies
-
Aneurysm vs Dissection
-
-
When to use deep hypothermic circulatory arrest (DHCA)
-
Removal of the Aorta
-
Sizing and placing the aortic graft
-
-
Modifications
-
Felt reinforcement
-
Anatomy
The aorta is the largest artery in the body to which all other major arteries are connected. It is a tube-like structure, usually about 20 - 30 mm in diameter, although its size varies proportionally to the height, weight, age and gender of the person. The aorta is widest at the point where it connects to the heart's left ventricle through the aortic valve; then, it gets progressively narrower as it descends into the abdomen.
Four Parts of the Aorta
-
Ascending aorta (figure 1)
-
Aortic arch (figure 2)
-
Thoracic Descending aorta (figure 3)
-
Abdominal aorta (figure 3)
The ascending aorta is composed of the initial attachment to the outflow tract from the heart beginning with the aortic root, aortic valve, sinuses of Valsalva, and sinotubular junction. The root is typically the largest part of the aorta. The ascending aorta continues via a slight curve towards the aortic arch.
The aortic arch bridges the ascending to the descending aorta. It typically contains 3 major branching vessels: the Innominate artery which divides into the right subclavian artery and the right common carotid artery, the left common carotid artery and the left subclavian artery.
Figure 1

Ascending Aorta

Arch
Innominate Artery
Right Subclavian Artery
Right Common Carotid Artery
Left Common Carotid Artery
Left Subclavian Artery
Figure 2
The descending thoracic aorta is a long, straight segment that runs from just below the left subclavian artery transitioning to the abdominal aortic aneurysm at the diaphragm eventually dividing into the left and right iliac arteries.

Descending Thoracic Aorta
Diaphragm
Abdominal Aorta
Common Iliac Artery
Figure 3
Just as the aortic arch has branch vessels the ascending and descending aorta also have significant branch vessels. The ascending aorta contains the left and right coronary arteries. The descending thoracic aorta contains bronchial, mediastinal, esophageal, pericardial, and superior phrenic arteries. The descending abdominal aorta contains Inferior phrenic, Celiac trunk, Superior and inferior mesenteric, Renal, gonadal, Lumbar, Median sacral, and Iliac arteries.
All blood vessels walls are composed of three layers. The tunica intima is the inner layer. This layer is a smooth surface where the blood flows on. It is thin and made of endothelial cells and supporting tissue. The tunica media is the middle layer. This layer is made of smooth muscle, elastic tissue, and collagen. The tunica adventitia is the outer layer. This layer is made of connective tissue like collagen and a network of small blood vessels (known as vasa vasorum) whose function is to nourish the aorta (figure 4).

Figure 4
Diseases of the Aorta - Aortic Aneurysm
Aneurysm of the ascending aorta usually result from degenerative changes in the aortic media including myxomatous changes or cystic medial degeneration often associated with connective tissue disorders such as Marfan disease, Ehlers-Danlos, polycystic kidney disease or a bicuspid aortic valve. Other common etiologies include post stenotic dilatation, infections such as endocarditis, mycotic aneurysms, trauma. Infections such as syphilis and salmonella. Other risk factors include age (>60), Male gender, family history, genetic factors, high cholesterol, hypertension. Smoking, diabetes, and obesity.
Aortic atherosclerosis is less common and are also degenerative in nature. Degenerative aneurysms are usually fusiform and may extend to the aortic annulus producing annulo-aortic ectasia resulting in valvular incompetence.
Aortic aneurysms are largely asymptomatic typically found incidentally on x-ray, CT or MRI performed for other reasons (figure 5). When symptoms do occur, it is usually a fatal or near fatal event related to rupture presenting as a severe tearing pain between the shoulder blades.

Figure 5
Diseases of the Aorta - Aortic Dissection
Aortic dissection is the surging of blood through a tear in the aortic intima with separation of the intima and media and creation of a false lumen or channel. The intimal tear may be a primary event or secondary to hemorrhage within the media. The dissection may occur anywhere along the aorta and extend proximally or distally into other arteries. Hypertension is an important contributor. Symptoms and signs include abrupt onset of tearing chest or back pain, and dissection may result in aortic regurgitation and compromised circulation in branch arteries. Diagnosis is by imaging tests including, transesophageal echocardiography, CT angiography, MRI, contrast aortography (figure 6).

Figure 6
Black patients, men, older patients, and patients with hypertension are especially at risk. Peak incidence occurs at age 50 to 65 years in the general population and at age 20 to 40 years for patients with congenital connective tissue disorders such as Marfan syndrome and Ehlers-Danlos syndrome.
Classification of Aortic Dissection
Aortic dissections are classified anatomically.
The DeBakey classification system is most widely used:
-
Type I (50% of dissections): These dissections start in the ascending aorta and extend at least to the aortic arch and sometimes beyond.
-
Type II (35%): These dissections start in and are confined to the ascending aorta (proximal to the brachiocephalic or innominate artery).
-
Type III (15%): These dissections start in the descending thoracic aorta just beyond the origin of the left subclavian artery and extend distally or, less commonly, proximally. Type IIIa dissections originate distal to the left subclavian artery and are confined to the thoracic aorta. Type IIIb dissections originate distal to the left subclavian artery and extend below the diaphragm. (figure 7)
The Stanford classification divides dissections by the most proximal involvement:
-
type A involves any part of the aorta proximal to the origin of the left subclavian artery (A affects ascending aorta)
-
type B arises distal to the left subclavian artery (figure 7)

Figure 7
A special case that is neither reflected in the original Stanford nor the DeBakey classification are dissections that involve the aortic arch but not the ascending aorta (between 8 and 15% of all aortic dissections). The nomenclature of these arch dissections has been incoherent for decades and still is.
American surgical consensus (2020) defines types A and B according to the location of the intimal tear (both types with additional qualifiers for proximal and distal extent):
-
type A: dissections with a tear in the ascending aorta including a segment with the branching of the brachiocephalic trunk
-
type B: all dissections with proximal tear distal to the branching of the brachiocephalic trunk
In contrast, a European surgical consensus document (2018) recognizes dissections of the arch without the involvement of the ascending aorta as a distinct category, termed "non-A-non-B dissection":
-
type A: proximal extent in ascending aorta
-
non-A-non-B dissection: retrograde extent or proximal tear in the arch between the brachiocephalic trunk and left subclavian artery
-
type B: proximal extent in descending aorta distal to left subclavian artery
-
A special case that is neither reflected in the original Stanford nor the DeBakey classification are dissections that involve the aortic arch but not the ascending aorta (between 8 and 15% of all aortic dissections 4). The nomenclature of these arch dissections has been incoherent for decades and still is.
Although dissection may originate anywhere along the aorta, it occurs most commonly at areas of greatest hydraulic stress, which are the
-
Right lateral wall of the ascending aorta (within 5 cm of the aortic valve)
-
Proximal segment of the descending aorta (just beyond the origin of the left subclavian artery)
Rarely, dissection is confined to individual arteries (eg, coronary or carotid arteries), typically in pregnant or postpartum women.
WHEN DO YOU OPERATE? (ACC/AHA Guidelines)
The following is an excerpt from the extensive ACC/AHA guidelines for management of aortic aneurysms.
Aortic Aneurysms, specific recommendations:
-
Non syndromic heritable thoracic aortic disease (nsHTAD). For patients with nsHTAD and no identifiable genetic cause, repair of the aorta is recommended when the diameter is ≥5.0 cm in the absence of high-risk features or ≥4.5 cm in the presence of high-risk features (family history of dissection at an aortic diameter <5.0 cm or unexplained sudden death at age <50 years, or rapid aortic growth).
-
Marfan syndrome (MFS).
-
Treatment is recommended with either a beta-blocker or an angiotensin receptor antagonist (ARB) in maximally tolerated doses.
-
Surgery to replace the aortic root and ascending aorta is recommended with an aortic root diameter ≥5.0 cm; surgery with an experienced surgeon is reasonable with either an aortic root diameter ≥4.5 cm plus high-risk features (family history of dissection, rapid growth, diffuse aortic root and ascending aorta dilation, or marked vertebral artery tortuosity), or with a cross-sectional aortic root area to patient height ratio ≥10 cm2/m.
-
-
Loeys-Dietz syndrome (LDS).
-
Treatment is reasonable with either a beta-blocker or an ARB in maximally tolerated doses.
-
Surgery for the prophylactic repair of the aortic root and ascending aorta should be based on the specific genetic variant, aortic diameter, aortic growth rate, extra-aortic features, family history, patient age and sex, and physician and patient preferences.
-
-
Bicuspid aortic valve (BAV) aortopathy.
-
All first-degree relatives should be screened with transthoracic echocardiography (TTE) for BAV and dilation of the aortic root and/or ascending aorta; computed tomography (CT) or magnetic resonance imaging (MRI) should be used if assessment with TTE is incomplete. It is reasonable to screen all first-degree relatives with TTE in the setting of a patient with BAV without aortopathy.
-
Surgery to replace the aortic root and/or ascending aorta is recommended with an aortic diameter ≥5.5 cm. Surgical intervention is reasonable with a cross-sectional area to height ratio ≥10 cm2/m, with an aortic diameter 5.0-5.4 cm plus a risk factor for dissection (family history of dissection, rapid growth, aortic coarctation, or "root phenotype" aortopathy), or with an aortic diameter ≥4.5 cm at the time of aortic valve replacement or repair.
-
-
Abdominal aortic aneurysm (AAA).
-
Ultrasound screening for AAA is recommended in men ≥65 years who have ever smoked and in men or women ≥65 years with a first-degree relative with AAA. Ultrasound screening is reasonable in women ≥65 years who have ever smoked.
-
Moderate- or high-intensity statin therapy is recommended in patients with AAA and evidence of aortic atherosclerosis. Low-dose aspirin may be considered in patients with AAA and concomitant atheroma or penetrating aortic ulcer (PAU).
-
Surveillance should be every 3 years in patients with AAA diameter 3.0-3.9 cm, annually in men with AAA diameter 4.0-4.9 cm or women with AAA diameter 4.0-4.4 cm, and every 6 months in men with AAA diameter ≥5.0 cm or women with AAA diameter ≥4.5 cm.
-
Repair is recommended in men with AAA diameter ≥5.5 cm or women with AAA diameter ≥5.0 cm, and in the setting of symptoms attributable to the aneurysm.
-
-
Sporadic/degenerative thoracic aortic aneurysm (TAA).
-
The use of antihypertensive medications is recommended among patients with sporadic TAA and blood pressure (BP) ≥130/80 mm Hg. In the absence of contraindications, the use of beta-blockers and/or ARB therapy is reasonable. Moderate- or high-intensity statin therapy is reasonable in patients with TAA and clinical or imaging evidence of atherosclerosis. Low-dose aspirin is reasonable in patients with TAA and concomitant atherosclerosis or PAU.
-
TTE is recommended at the time of diagnosis to assess aortic valve anatomy and function and thoracic aortic diameters. CT or MRI is reasonable at the time of diagnosis. Surveillance imaging with TTE, CT, or MRI is reasonable after 6-12 months, and then, if stable, every 6-24 months.
-
For aortic root or ascending aorta aneurysm, surgery is recommended for symptoms attributable to the aneurysm, diameter ≥5.5 cm, or rapid growth; and reasonable at the time of tricuspid aortic valve replacement with an aortic diameter ≥5.0 cm. With an experienced surgeon, surgery is reasonable with an aortic diameter ≥5.0 cm (or in a patient with height >1 standard deviation above or below the mean with a cross-sectional aortic area to height ratio ≥10 cm2/m), or at the time of tricuspid aortic valve replacement with an aortic diameter ≥4.5 cm.
-
Thoracic endovascular aortic repair (TEVAR) is recommended over open surgery in patients without MFS, LDS, or vascular Ehlers-Danlos syndrome who have descending TAA meeting criteria for intervention and suitable anatomy; consideration for alternative vascular access is recommended for patients with smaller or diseased access vessels.
-
-
Surveillance after TAA repair.
-
After treatment of TAA with TEVAR, surveillance imaging with CT is recommended after 1 month and 12 months, and then annually if stable; MRI is a reasonable alternative to CT.
-
After open repair of TAA, CT or MRI within 1 year and then every 5 years is reasonable in the absence of residual aortopathy; annual imaging is reasonable if there is residual aortopathy or abnormal findings on surveillance imaging.
-
Aortic Dissection, specific recommendations:
Acute type A aortic dissection is a life-threatening condition because of potential sequelae, including rupture that causes cardiac tamponade, acute severe aortic regurgitation that causes heart failure or shock, compromised coronary artery ostia causing myocardial ischemia, or malperfusion causing end-organ ischemia or infarction, all of which can all be fatal. Suspected or diagnosed acute type A aortic dissection warrants urgent surgical evaluation, because the mortality rate of medical management alone is 2 to 3 times that of surgical intervention.
1. Data from IRAD showed that from 1995 to 2013, the surgical mortality rate decreased from 25% to 18%, while the medical mortality rate remained unchanged at 57%. Surgical intervention mitigates the immediate risk of aortic rupture/tamponade, corrects aortic valve regurgitation and myocardial ischemia, and reestablishes flow to malperfused vessels. Although age is a risk factor, elderly patients still benefit from surgery, with superior immediate and midterm outcomes compared with medical therapy. Short and midterm outcomes can be equivalent to younger populations, with circulatory collapse being the primary predictor of long-term survival. In patients with significant contraindications to surgery, including frailty, clinical judgment may determine that the risk-benefit ratio favors medical management. The potential sequelae of acute type A aortic dissection, including myocardial infarction, acute AR, cardiac tamponade, aortic rupture, and end-organ malperfusion, are associated with high rates of morbidity and mortality. Given the acuity, unpredictability, and finality of such events, immediate evaluation for surgical intervention is warranted to reverse any ongoing physiologic compromise and mitigate the risk of fatal events. The mortality rate of unoperated acute type A aortic dissection is 1%/h, and the time intervals between symptom onset, diagnosis, and surgery have a significant effect, with the highest mortality rate occurring in those undergoing surgery 8 to 12 hours after diagnosis. Patients presenting with clinical indicators of severe physiologic compromise (shock, neurologic deficits, malperfusion, myocardial ischemia) mandate the most immediate consideration for repair as the only potential option for survival.
2. Patients with acute type A aortic dissection who present with hemodynamic stability have an unpredictable course because of the inability to predict eventual rupture. Although some studies have suggested that night-time surgery is associated with a higher mortality rate,17,18 other studies have shown no diurnal difference in outcomes, and all studies have shown no difference with weekend surgery. Surgeon and center experience and resource availability should be considered to ensure optimal outcomes. Despite an inherent delay in the start time of surgery, transfer from low to high-volume hospitals (one that performs ≥7 aortic root, ascending aorta, or transverse arch aortic dissection repairs per year), as part of regionalization of care, can result in significantly improved outcomes.
3. In patients with cerebral malperfusion, survival is superior with surgery; in patients with acute type A aortic dissection and an acute stroke, the mortality rates of surgical versus medical management are 25% to 27% versus 76%, respectively. Even more strikingly, Estrera et al showed that patients with acute type A aortic dissection who had presented with stroke had an operative mortality rate of only 7% and showed no worsening of neurologic status postoperatively. Although their study and others, have emphasized the timeliness of the aortic repair in stroke patients, with a cutoff of ∼5 to 10 hours (after which neurologic outcomes declined), Fischbein et al found no association between postoperative neurologic improvement and time from onset of neurologic symptoms to surgery. IRAD data revealed that cerebrovascular accident and coma resolved in 84% and 79% of patients, respectively, despite mean times to surgery of 12.3 and 13.8 hours, respectively. It should be noted, however, that in 1 recent report of 11 patients with acute type A aortic dissection and complete occlusion of an internal carotid artery, all died from cerebral edema and herniation, regard less of management; consequently, this particular subset of patients may not benefit from surgical intervention.
The Procedure - Replacement of Ascending Aorta


Repair for aortic aneurysm and aortic dissection are fairly similar with a few acceptations primarily referring to the site of anastomosis and cannulation strategies. Before getting into the specific procedure, I would like to discuss cannulation strategies.
When we think of cardiac surgical procedures the need for end organ protection comes to mind. A majority of cardiac surgical procedures require cardiopulmonary bypass. This requires placement of cannulas that innervate the cardiopulmonary bypass machine to support end organ perfusion. Although routine aortic and venous cannulation occurs with some aortic surgery there are circumstances that preclude the use of direct aortic cannulation. This typically involves aortic dissection cases where there are concerns for cannulating the false vs true lumen and the potential for acute rupture when manipulating the aorta. Therefore, alternative modes of cannulation and perfusion are required such as femoral or axillary artery cannulation (figure 1).
Another consideration is the need to perform the anastomosis near the aortic arch. In these cases, the patient will need to be cooled to 18-20 degrees centigrade so the bypass machine can be stopped in a process we call deep hypothermic circulatory arrest (DHCA). When blood circulation stops at normal body temperature (37 °C), permanent damage occurs in only a few minutes. More damage occurs after circulation is restored. Reducing body temperature extends the time interval that such stoppage can be survived. At a brain temperature of 14 °C, blood circulation can be safely stopped for 30 to 40 minutes. There is an increased incidence of brain injury at times longer than 40 minutes, but sometimes circulatory arrest for up to 60 minutes is used if life-saving surgery requires it.
At our institution a majority of aortic aneurisms are cannulated in the usual fashion via the aorta. If the aneurysm extends to the aortic arch, DHCA will be required. In this circumstance additional limbs may be added to the aortic tubing from the bypass circuit to include cannulas for perfusing the head vessels for antigrade cerebral perfusion (figure 2)


Figure 1: Axillary arterial cannulation on the left, femoral arterial and venous cannulation on the right.

Figure 2: image on the left: CPB tubing configuration for antegrade cerebral perfusion. Image on the right: Placement of cannula for antegrade cerebral perfusion.

The most straight forward aortic replacement is addressing a localized aneurysm that involves the aorta just above the sinotubular junction to a site below the aortic arch that allows space for aortic cannulation, aortic cross clamp and an adequate rim of tissue for the anastomosis (figure 8a,b,c,d). If these three things are not available, the patient will need to have DHCA in order to perform the distal anastomosis adjacent to the aortic arch. If the patient requires DHCA the patient will start to be cooled as soon as CPB is instituted.

Figure 8a

Figure 8c

Figure 8b

Figure 8d
Prior to cross clamping the aorta is separated from the pulmonary artery and other connective tissue under the vessel (figure 9a&b). If it has been determined that the anastomosis can be completed without DHCA, the aorta is cross clamped, and heart is arrested in the usual standard fashion. During this process the assistant helps with the dissection between the aorta and pulmonary artery by grasping and retracting the tissue on the PA side of the aorta using a DeBakey forceps while the surgeon retracts the aortic side. You may also need suction available to keep the area dry. The suction also helps with retraction of the tissue (figure 9a). To assist with dissection of the posterior table behind the aorta and just above the right pulmonary artery the assistant retracts the aorta using either a peanut or as in this case your hand (figure 9b).

Figure 9a

Figure 9b
If the patient requires concomitant procedures for valvopathy such as, aortic valve stenosis or regurgitation, the valve will be addressed while the patient is being cooled for DHCA. Once the heart is arrested the aneurysmal section of the aorta is resected at the at the proximal end first. It is opened similar to the incision used for and aortic valve replacement just above the sinotubular junction (figure 10) the incision is the extended laterally in both directions. Assisting with the process includes the use of a DeBakey forceps to retract the epicardial fat and suction to keep the area dry (figure 10).

Figure 10
Additional dissection of connective tissue near the pulmonary artery and posteriorly is completed. During this process the assistant helps by retracting the tissue opposite the surgeon to expose the space. The use of a fine tip suction is useful in keeping the field dry while keeping a low profile so as not to interfere with the process (figure 11).

Fine tip suction
Figure 11
A retention suture is placed and the aorta is retracted up towards the head creating space for the proximal anastomosis (figure 12).

Figure 12
THE DISTAL ANASTIMOSIS
We use a Hemashield graft for our aortic replacements. It is made of woven polyester fabric coated with a highly purified form of cross-linked Type I bovine collagen. It is designed to reduce bleeding at implant and gradually be reabsorbed by the patient. This particular graft has a branch graft that will be used for reattaching the cardiopulmonary circuit (figure 13).

Branch graft
Figure 13
In terms of location the proximal anastomosis is at the sinotubular junction whereas the distal anastomosis is toward the aortic arch. The process for sewing the proximal and distal anastomosis it the same. Typically starting from the most inferior portion and proceeding clockwise or counterclockwise according to the surgeon's preference. In this discussion I will cover replacing the ascending aortic replacement with circulatory arrest.
As described previously, deep hypothermic circulatory arrest (DHCA) is performed when a portion of the aneurysm is too close to the arch to perform the distal anastomosis. This requires cooling the patient to 20 degrees for end organ protection. In an effort to maintain good cerebral protection a cannula will be placed in the innominate artery to the right carotid artery. During circulatory arrest the brain will be perfused using antigrade cerebral perfusion.
While the patient is being cooled the surgeon may turn attention to the proximal anastomosis, however, in this particular case the aortic valve was replaced as well. During that time the patient was cooled sufficiently to circ arrest and complete the distal anastomosis first.
While instituting DHCA the cross clamp and aortic cannula are removed. to trim back the remanent of the ascending aortic aneurysm (figure 14a & b).

Figure 14a

Figure 14b
Following institution of DHCA the aorta is trimmed to just beyond the aneurismal section. During this process it is the assistant's responsibility to keep the field dry and apply counter traction to expose tissue that needs to be resected (figure 15 a-c).
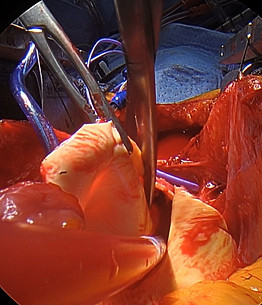
Figure 15a
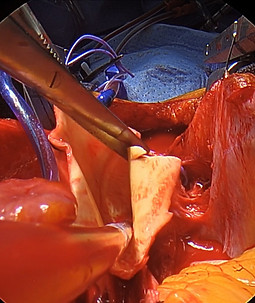
Figure 15b

Figure 15c
With the aortic resection complete and just prior to beginning the anastomosis the surgeon will need to place the cannula for antegrade cerebral perfusion (ACP). In this image the field is dry due to the placement of a sump sucker attached to the pump sucker tubing. This will return all bleeding to the pump to be recirculated (figure 16). Once the cannula is in place a tourniquet it tightened, and ballon inflated. Antegrade flow should be seen returning via the left common carotid confirming an intact Circle of Willis. Some surgeons may have a second cannula available to perfuse the left common carotid if this is not observed.

ACP Cannula
Sump sucker
Figure 16
With the aorta trimmed to size and the ACP cannula in place the surgeon can now begin the anastomosis starting with the posterior wall. Note that the graft is away from the aorta to start so the surgeon can place the posterior sutures precisely. Another important thing to notice is the use of a felt strip. A characteristic of aneurysmal tissue is that it is thinned out. A felt strip is used to reenforce the anastomosis. The anastomosis is started slightly to the assistant's side. This technique will give the surgeon good exposure to the complete posterior aspect of the anastomosis. Once the anastomosis is complete that particular area is difficult to access if a repair is needed (figure 16 a & b). As the assistant you will be responsible for following the suture, maintaining a dry field and retracting the graft for exposure.

Figure 16a

Figure 16b
Following the first three to four bites in the graft and aorta the graft is parachuted down (figure 17).

Figure 17
The anastomosis iw continued toward the surgeon in a clockwise manner (figure 18 a & b). After passing the posterior and partially up the surgeon side of the anastomosis some surgeons will opt to place two to three pledgeted reinforcement sutures in the posterior aspect of the anastomosis. As mentioned above, the posterior aspect is the most difficult to repair so placing these sutures is just one more extra level of support (figure 18 c & d).

Figure 18a

Figure 18c

Figure 18b

Figure 18d
As you can see from the images the field is fairly dry. This frees up both hands of the assistant to follow the suture and if necessary, move cannulas such as the sump and ACP, out of out of the way as in figure 18c, the assistant is moving the sump aside so the surgeon can complete the reinforcement suture.
TIP: When following the suture remember to maintain a 50/50 distance. This will avoid the potential for knot formation or locking of the suture (figure 19). The formation of a knot can make it difficult to complete the anastomosis as it will not smoothly go through tissue and almost never will go through a graft.

Figure 19 Knot noticed before suture was placed allowing the surgeon to unravel the suture.
The surgeon will continue up the side nearest them to the point where the graft starts to curve to the anterior portion of the anastomosis (figure 20a). They will then proceed to the opposite side of the anastomosis sewing from the posterior lateral area, along the lateral wall to the anterior portion of the graft (figure 20b & c). During this process the assistant will continue to follow the suture, keeping the anastomosis tight with each bite.
TIP: One of the most common and avoidable causes of bleeding is a loose suture line. You can also help by pulling the graft back to facilitate suture placement as the surgeon reaches the anterior portion of the anastomosis. Graft/Aortic miss match is another important concept to be aware of. The graft measurement is not always exact. Quite often the surgeon will need to make some adjustments along the anastomosis to make up for differences in size relative to the aorta or graft (figure 21a & b).
These adjustments involve taking a larger or smaller bite in one vs the other. For instance, if the graft is slightly larger than the aorta, the surgeon will take a slightly larger bite or advance in the graft than the aorta. Over the distance of the entire anastomosis the difference will be made up. These adjustment bites occur at the from the lateral aspects to the anterior portion of the anastomosis. Because some of these adjustments may result in small gaps that may require repairs they are never made in the posterior aspect of the anastomosis. While the anastomosis is being performed both the surgeon, and the assistant are assessing the size match. If there is a difference it is incumbent upon the assistant to mention the discrepancy if it appears the surgeon has not begun to compensate. There are times the assistant may notice this before the surgeon. The sooner the issue is recognize the easier it is to correct.
As the anastomosis nears completion at the left anterolateral location, the ACP cannula will be removed, and the anastomosis finished (figure 22a & b).



Figure 20c
Figure 20b
Figure 20a


Figure 21a
Figure 21b

Figure 22a

Figure 22b
While the surgeon is completing the anastomosis the assistant is maintaining a tight suture line with the follow and when needed, retracting the graft.
TIP: When following at the end of the anastomosis you may need to relax on the follow so the surgeon can see the edge of the aorta so as not the get a partial thickness bite in the aorta. With each bit into through the aorta the assistant will then pull the suture tight. This process my need to be repeated 3 or 4 times depending in the length of anastomosis remaining. As the surgeon is tying the suture the assistant can be getting the aortic line and graft connected in preparation for reinstituting CPB (figure 23).
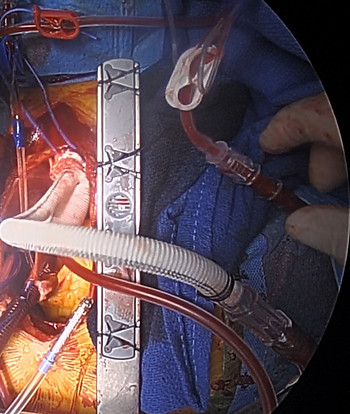
Figure 23
With the anastomosis complete the process of reinstituting cardiopulmonary bypass (CPB) can begin. If DHCA was used, following reinstitution of CPB the patient is rewarmed. The first step is to deair the graft prior to cross clamping. This is performed by maintaining Trendelenburg that was done during DHCA and slowly starting CPB with the graft open to the field. Blood will begin flowing out of the grafts the surgeon shakes the graft to liberate air trapped in the graft and rises out of the aorta (figure 24a). The graft is then clamped and full CPB resumed (figure 24b).
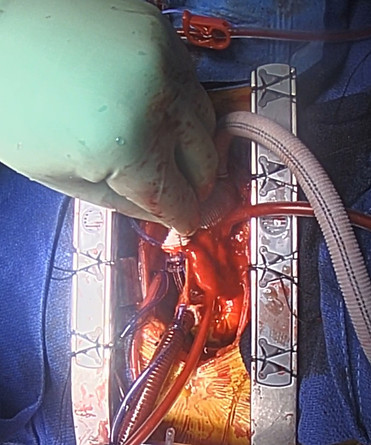
Figure 24a
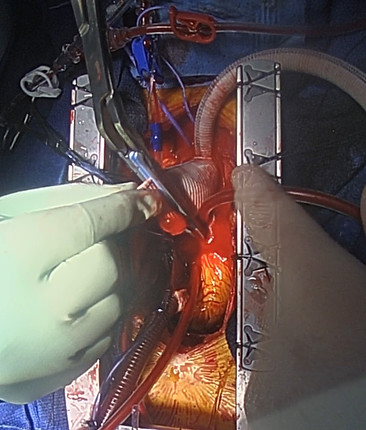
Figure 24b
In figure 24b Notice that the cross clamp is very close to the potential anastomosis. Hemashield grafts have an accordion-like design that expands when pressurized. Due to this property, it is important to stretch the graft to fit the correct length. If the anastomosis is completed without sizing the graft properly it may be too long resulting in kinking of the graft. In addition to creating the proper length the surgeon also needs to develop the correct shape. The normal curve of the aorta is and arc with the short side posteriorly and toward the pulmonary artery. This needs to be part of the sizing process. By filling the graft under pressure, it will expand to the final size creating a more physiologic sizing process. With the graft expanded the surgeon can measure and mark the anticipated length (figure 25a). The cross clamp is then moved back to afford room to shorten and sew the graft (figure 25b).

Figure 25a

Figure 25b
The graft is cut to the predetermined size and proximal anastomosis started (figure 26).

Figure 26
THE PROXIMAL ANASTIMOSIS
The process for sewing proximal anastomosis is nearly the same as the distal. The main distinction is the lack of mobility with the graft. The graft is anchored distally so there is less movement available for exposure. The anastomosis is beveled to create the contour needed for simulating the anatomic position of the graft similar to the native position. This beveling will create a larger graft to aorta mismatch that will need to be adjusted for. With that in mind the anastomosis is started in a similar fashion as the distal using a circumferential felt strip for reinforcement (figure 27a). The first bite is made slightly to the assistant's side with the graft and aorta slightly apart so the surgeon can complete the first 2-3 bites with precision (figure 27b). As the assistant the main duty is exposure for each bite and suture follow. What you do will vary depending on the needs of the surgeon such as assisting with felt strip placement, landing of suture loops to maintain proper spacing, or, as in figure 27b, retracting the graft using a DeBakey forceps to expose the aorta and graft edges with your right hand and following the suture with the left.

Figure 27a

Figure 27b
In this case the aorta and graft are approximated after the first two bites. The anastomosis continues along the posterior aspect and surgeon's lateral side (figure 28a & b.

Figure 28a

Figure 28b
As with the distal anastomosis, once the surgeon has completed part of the lateral anastomosis, two to three pledgeted support sutures may be placed at the posterior aspect of repair (figure 29).


Figure 29 Support stitch to the posterior aspect of the anastomosis. On the left the surgeon is placing the second bite while the assistant is placing the suture through the free pledget. On the right the first support stich has been placed.
TIP: There are ways, as an assistant, to facilitate the processes as described above. Another important concept is suture placement. It is important to have a consistent suture placement along the anastomosis. The assistant can help by guiding the suture as it is placed. Notice in figure 30, the space between each suture is even with no gaps. As the surgeon continues with the anastomosis the assistant needs to pay attention to the suture spacing. If there is an uneven line with a significant gap there will be an opportunity for a leak through the anastomosis

Figure 30
Suture manipulation can also facilitate the anastomosis. For example, holding the follow away from the direction the needle will be coming through the tissue or graft.

Suture direction
Follow direction

Follow direction
Suture direction
Figure 31 In the image on the left the needle is coming through the aorta toward the surgeon with the follow held toward the head. On the right the needle is coming through the graft towards the assistant and the follow is held toward the feet.
Small adjustments like this can prevent locking the stitch and helps to facilitate the process in a more efficient fashion.
Following the placement of the reinforcement stitches, the surgeon will continue up the surgeon side until just below the curve to the anterior aspect of the anastomosis (figure 32a). Once the curve to the anterior aspect has been reach the surgeon will switch to the assistant side of the anastomosis. The anastomosis will be completed in this clockwise direction (figure 32b).

Follow
Needle
Needle Driver Through Loop
Figure 32a

Follow
Needle
Figure 32b
Note a few things in the above images. In figure 32a the follow is toward the assistant slightly toward the head with the needle coming out of the aorta towards the surgeon. Another interesting thing to point out is the needle holder through the loop in the stitch. If that goes unnoticed it can result in a knot. In image 32b the follow is toward the feet, the needle is coming out through the graft toward the assistant. The surgeon will continue up the assistant side in a clockwise fashion until reaching the suture on the opposite side (figure 33a & b).

Figure 33a

Figure 33b
The space becomes smaller as the surgeon approaches the apex of the anastomosis. It is very important that the surgeon is able to see the edge of the aorta and the graft in order to get a full thickness bite. The assistant can help facilitate this by relaxing on the follow while retracting the graft with each bite (figure 34a & b).

Figure 34a

Figure 34b
THE PROXIMAL ANASTIMOSIS
The process for sewing proximal anastomosis is nearly the same as the distal. The main distinction is the lack of mobility with the graft. The graft is anchored distally so there is less movement available for exposure. The anastomosis is beveled to create the contour needed for simulating the anatomic position of the graft similar to the native position. This beveling will create a larger graft to aorta mismatch that will need to be adjusted for. With that in mind the anastomosis is started in a similar fashion as the distal using a circumferential felt strip for reinforcement (figure 27a). The first bite is made slightly to the assistant's side with the graft and aorta slightly apart so the surgeon can complete the first 2-3 bites with precision (figure 27b). As the assistant the main duty is exposure for each bite and suture follow. What you do will vary depending on the needs of the surgeon such as assisting with felt strip placement, landing of suture loops to maintain proper spacing, or, as in figure 27b, retracting the graft using a DeBakey forceps to expose the aorta and graft edges with your right hand and following the suture with the left.

Figure 27a

Figure 27b
In this case the aorta and graft are approximated after the first two bites. The anastomosis continues along the posterior aspect and surgeon's lateral side (figure 28a & b.

Figure 28a

Figure 28b
As with the distal anastomosis, once the surgeon has completed part of the lateral anastomosis, two to three pledgeted support sutures may be placed at the posterior aspect of repair (figure 29).


Figure 29 Support stitch to the posterior aspect of the anastomosis. On the left the surgeon is placing the second bite while the assistant is placing the suture through the free pledget. On the right the first support stich has been placed.
TIP: There are ways, as an assistant, to facilitate the processes as described above. Another important concept is suture placement. It is important to have a consistent suture placement along the anastomosis. The assistant can help by guiding the suture as it is placed. Notice in figure 30, the space between each suture is even with no gaps. As the surgeon continues with the anastomosis the assistant needs to pay attention to the suture spacing. If there is an uneven line with a significant gap there will be an opportunity for a leak through the anastomosis

Figure 30
Suture manipulation can also facilitate the anastomosis. For example, holding the follow away from the direction the needle will be coming through the tissue or graft.

Suture direction
Follow direction

Follow direction
Suture direction
Figure 31 In the image on the left the needle is coming through the aorta toward the surgeon with the follow held toward the head. On the right the needle is coming through the graft towards the assistant and the follow is held toward the feet.
Small adjustments like this can prevent locking the stitch and helps to facilitate the process in a more efficient fashion.
Following the placement of the reinforcement stitches, the surgeon will continue up the surgeon side until just below the curve to the anterior aspect of the anastomosis (figure 32a). Once the curve to the anterior aspect has been reach the surgeon will switch to the assistant side of the anastomosis. The anastomosis will be completed in this clockwise direction (figure 32b).

Follow
Needle
Needle Driver Through Loop
Figure 32a

Follow
Needle
Figure 32b
Note a few things in the above images. In figure 32a the follow is toward the assistant slightly toward the head with the needle coming out of the aorta towards the surgeon. Another interesting thing to point out is the needle holder through the loop in the stitch. If that goes unnoticed it can result in a knot. In image 32b the follow is toward the feet, the needle is coming out through the graft toward the assistant. The surgeon will continue up the assistant side in a clockwise fashion until reaching the suture on the opposite side (figure 33a & b).

Figure 33a

Figure 33b
The space becomes smaller as the surgeon approaches the apex of the anastomosis. It is very important that the surgeon is able to see the edge of the aorta and the graft in order to get a full thickness bite. The assistant can help facilitate this by relaxing on the follow while retracting the graft with each bite (figure 34a & b).

Figure 34a

Figure 34b
THE PROXIMAL ANASTIMOSIS
The process for sewing proximal anastomosis is nearly the same as the distal. The main distinction is the lack of mobility with the graft. The graft is anchored distally so there is less movement available for exposure. The anastomosis is beveled to create the contour needed for simulating the anatomic position of the graft similar to the native position. This beveling will create a larger graft to aorta mismatch that will need to be adjusted for. With that in mind the anastomosis is started in a similar fashion as the distal using a circumferential felt strip for reinforcement (figure 27a). The first bite is made slightly to the assistant's side with the graft and aorta slightly apart so the surgeon can complete the first 2-3 bites with precision (figure 27b). As the assistant the main duty is exposure for each bite and suture follow. What you do will vary depending on the needs of the surgeon such as assisting with felt strip placement, landing of suture loops to maintain proper spacing, or, as in figure 27b, retracting the graft using a DeBakey forceps to expose the aorta and graft edges with your right hand and following the suture with the left.

Figure 27a

Figure 27b
In this case the aorta and graft are approximated after the first two bites. The anastomosis continues along the posterior aspect and surgeon's lateral side (figure 28a & b.

Figure 28a

Figure 28b
As with the distal anastomosis, once the surgeon has completed part of the lateral anastomosis, two to three pledgeted support sutures may be placed at the posterior aspect of repair (figure 29).


Figure 29 Support stitch to the posterior aspect of the anastomosis. On the left the surgeon is placing the second bite while the assistant is placing the suture through the free pledget. On the right the first support stich has been placed.
TIP: There are ways, as an assistant, to facilitate the processes as described above. Another important concept is suture placement. It is important to have a consistent suture placement along the anastomosis. The assistant can help by guiding the suture as it is placed. Notice in figure 30, the space between each suture is even with no gaps. As the surgeon continues with the anastomosis the assistant needs to pay attention to the suture spacing. If there is an uneven line with a significant gap there will be an opportunity for a leak through the anastomosis

Figure 30
Suture manipulation can also facilitate the anastomosis. For example, holding the follow away from the direction the needle will be coming through the tissue or graft.

Suture direction
Follow direction

Follow direction
Suture direction
Figure 31 In the image on the left the needle is coming through the aorta toward the surgeon with the follow held toward the head. On the right the needle is coming through the graft towards the assistant and the follow is held toward the feet.
Small adjustments like this can prevent locking the stitch and helps to facilitate the process in a more efficient fashion.
Following the placement of the reinforcement stitches, the surgeon will continue up the surgeon side until just below the curve to the anterior aspect of the anastomosis (figure 32a). Once the curve to the anterior aspect has been reach the surgeon will switch to the assistant side of the anastomosis. The anastomosis will be completed in this clockwise direction (figure 32b).

Follow
Needle
Needle Driver Through Loop
Figure 32a

Follow
Needle
Figure 32b
Note a few things in the above images. In figure 32a the follow is toward the assistant slightly toward the head with the needle coming out of the aorta towards the surgeon. Another interesting thing to point out is the needle holder through the loop in the stitch. If that goes unnoticed it can result in a knot. In image 32b the follow is toward the feet, the needle is coming out through the graft toward the assistant. The surgeon will continue up the assistant side in a clockwise fashion until reaching the suture on the opposite side (figure 33a & b).

Figure 33a

Figure 33b
The space becomes smaller as the surgeon approaches the apex of the anastomosis. It is very important that the surgeon is able to see the edge of the aorta and the graft in order to get a full thickness bite. The assistant can help facilitate this by relaxing on the follow while retracting the graft with each bite (figure 34a & b).

Figure 34a

Figure 34b
With the anastomosis complete the surgeon will turn attention to removing the cross-clamp, continued rewarming if needed, deairing the heart and repairing leaks in the aorta-graft interface prior to coming off CPB. Once the patient comes of CPB and the heart is full it becomes problematic to repair had to reach locations such the posterior aspect of the repair at the proximal and distal ends. Prior to removal of the cross-clamp an antigrade cannula will be places to assist with deairing of the heart (figure 35a). Once the patient has been rewarmed and major bleeding issues addressed the clamp is removed and the patient is weaned from bypass in the usual fashion (fighure 35b).

Figure 35a

Figure 35b
Once the patient has been weaned off CPB, hemodynamically stable, and heparin has been reversed decannulation can proceed in the usual fashion. The only difference in this case and others that require alternative cannulation techniques that create slight variations. In this particular case the graft limb will be ligated, clipped with extra-large clips and oversewn near the base of the junction indicated by the arrow in figure 35b. Other possible sites that require repair as part of decannulation include axillary artery cannulation, often used in aortic dissections, and femoral artery repair if femoral access was needed. For details on decannulation to closure please refer to the Laying the foundation observation page.
